Some diseases are well understood, with clear causes and established treatments. Others remain unresolved, sitting at the edge of what we know. Canine dysautonomia (CD) is one of those diseases. As a veterinarian and PhD student at the University of Wyoming, my work focuses on understanding canine dysautonomia where multiple systems—and possibly multiple factors— interact.
Dysautonomia Across Veterinary Species
Canine dysautonomia is a severe neurodegenerative disorder. It affects the autonomic nervous system (ANS), both sympathetic and parasympathetic divisions. The ANS controls essential involuntary functions such as gastrointestinal motility, heart rate, urination, and pupil response.
When this system fails, the clinical effects are widespread and often devastating.
Affected dogs typically present with a combination of signs reflecting generalized autonomic failure. Common clinical features include gastrointestinal dysfunction (ileus, regurgitation, constipation), dysphagia, dilated pupils with poor or absent light response, reduced tear and nasal secretions, urinary retention or incontinence, and generalized weakness. Cardiovascular abnormalities may also be present, including little or no change in heart rate following mild exercise or atropine administration.
One useful clinical test is the pilocarpine response test, in which clinicians apply dilute pilocarpine to the eye. Dogs with dysautonomia show rapid pupil constriction (miosis) due to denervation hypersensitivity, a finding that can help support the diagnosis in the appropriate clinical context.
Despite its severity, the underlying cause remains unknown.
Canine dysautonomia does not occur in isolation. Researchers have described similar diseases affecting the autonomic nervous system in multiple species, including horses, cats, rabbits, sheep, and even birds. In horses (equine dysautonomia or grass sickness), cats (feline dysautonomia), and dogs, the disease typically presents with acute onset, widespread autonomic dysfunction, and non-inflammatory neurodegeneration. Veterinary pathologists have also reported similar lesions in rabbits and sheep, although less commonly.
Although there are differences between mammalian species—likely related to host susceptibility, environmental exposure, and specific causative agents—the overall similarities in clinical presentation, pathology, and epidemiological patterns suggest that these conditions may share a common or related underlying cause, possibly involving environmental factors and toxin-mediated injury to autonomic neurons.
Geographic Distribution of Canine Dysautonomia
Canine dysautonomia shows a distinct and non-random geographic distribution that veterinarians have recognized since clinicians first reported it in England in 1983. Following the initial cases in the United Kingdom, additional reports emerged across parts of Europe, including Scotland, Belgium, and Norway. Researchers later identified the disease in the United States in 1991 and have since reported it in multiple regions of North America.
Within the United States, cases tend to cluster in the Midwestern region, particularly in states such as Missouri and Kansas, with additional reports from areas including Colorado, Wyoming, and surrounding regions. Beyond the U.S., scientists have also described cases in Canada and parts of Europe, reinforcing that this is not a regionally isolated condition.
What makes this pattern especially notable is that cases occur as sporadic incidents or small clusters, rather than spreading between animals. This global yet localized distribution strongly suggests that environmental exposure—rather than contagious transmission—plays a central role in the disease.
What Do We Know So Far? (Epidemiological Clues)
Although the exact cause of CD is unclear, several consistent epidemiological patterns have emerged:
- Many affected dogs have a history of recent soil exposure or digging behavior (Figure 1)
- The disease is not considered contagious as households of several dogs may only have one sick dog.
- Cases often occur in puppies, median age 18 months, with access to rural property
- Cases tend to appear as isolated incidents or small clusters (dogs visiting same disturbed area)
- A seasonal pattern is observed, with peaks in late winter and early spring
Together, these patterns have led to the hypothesis that soil-associated microbes or toxins may cause the disease. Many affected dogs (45%? Add the correct stat here) have a history of digging or close contact with disturbed soil prior to disease onset. However, identifying a specific agent has proven difficult.
What Happens at the Tissue Level?
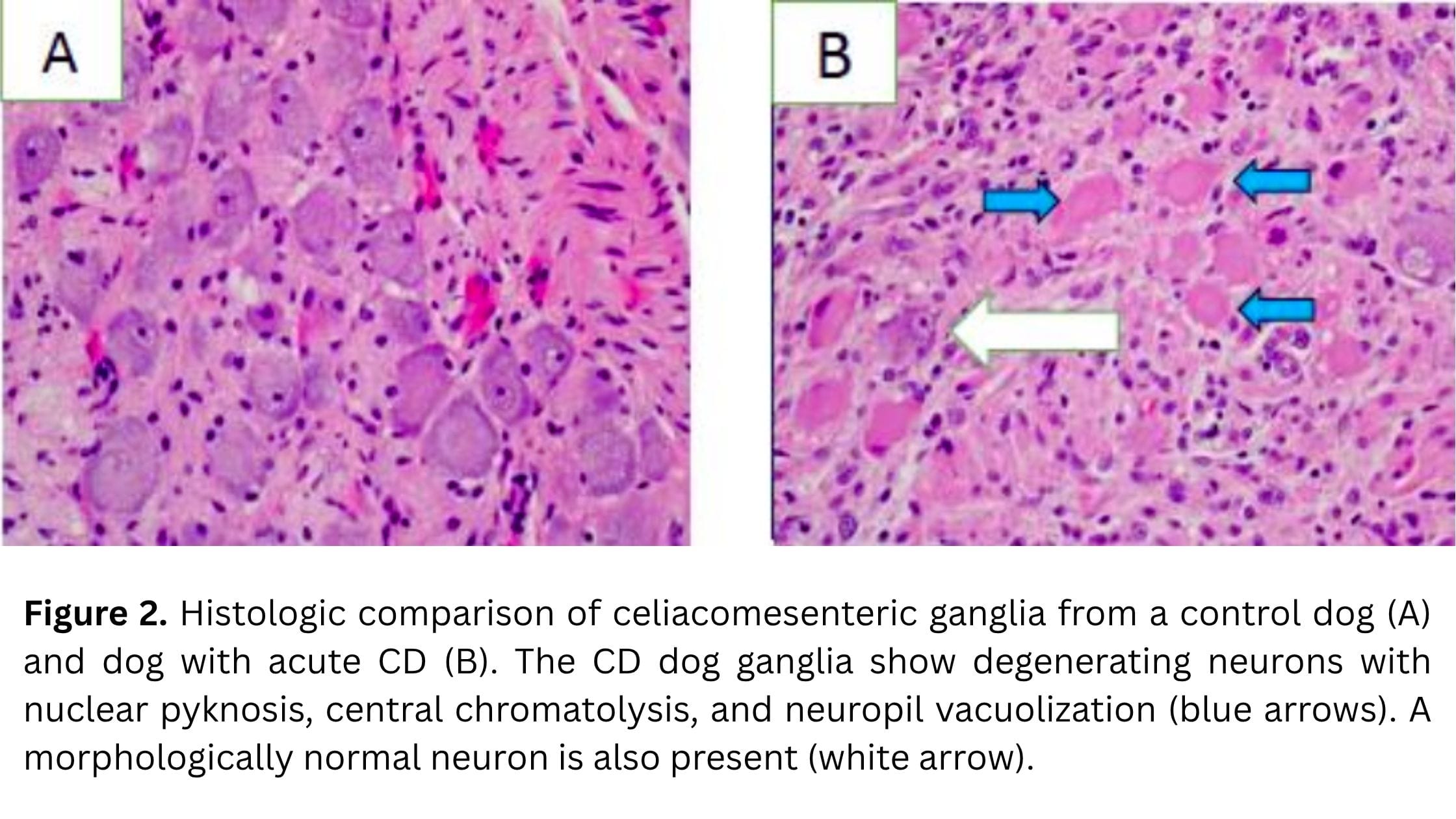
Histopathological findings provide important insight into the disease process. The hallmark lesion in canine dysautonomia is degeneration of neurons within autonomic ganglia. Affected neurons commonly show chromatolysis, characterized by cell body swelling, loss of Nissl substance, and displacement of the nucleus. In more advanced cases, neuronal necrosis and loss is evident (Figure 2).
There is often minimal to mild inflammation, which supports the idea that the process is not primarily inflammatory. Instead, the pattern of injury suggests a toxic or metabolic insult targeting autonomic neurons. These changes are widespread and consistent with the diffuse autonomic dysfunction observed clinically, but what triggers this damage remains the central question.
A Multi-Disciplinary Approach to Understanding CD
To address this complexity, our research group at the University of Wyoming—led by Dr. Jonathan Fox (Principal Investigator) and Dr. Alison Doherty (Co-Principal Investigator)—is taking an integrated approach. Rather than focusing on a single hypothesis, we posited three aims to understand canine dysautonomia from a broader perspective—one that considers the possible interaction between environmental exposure, microbes, and the host response. Given the consistent association of the disease with soil exposure and outdoor environments, one central question is there a specific microbe, or a toxin-encoding gene, associated with canine dysautonomia? To explore this, we carried out genomic work using fecal samples collected from dogs diagnosed with CD, along with healthy controls. We compared microbial and genetic profiles with the goal of identifying a potential microbial or toxin-related component of the disease absent in controls.
However, identifying genetic material alone is not enough. A key question is whether these potential toxin-related elements are biologically active. For this reason, we also incorporate proteomic approaches to examine whether relevant proteins are expressed, helping us move from what could be present to what may be functionally important.
Finally, we study the disease from the host side using metabolomics. By analyzing small molecules in plasma samples, we aim to understand how the disease alters normal physiological pathways. This helps provide insight into the mechanisms underlying autonomic dysfunction, rather than focusing on a single cause.
By combining microbial, proteomic, and metabolomic perspectives, we aim to build a more complete understanding of how these components interact with the goal of finding the cause of CD. The research is expensive, and we are grateful to the American Kennel Club Canine Health Foundation and individual donors who support our work.
Looking Ahead
Although canine dysautonomia remains an unresolved disease, ongoing research continues to move the field forward Improving our understanding could eventually lead to better diagnostic tools, earlier detection, and more targeted therapeutic strategies. While those advances may still be some distance away, the long-term goal is clear: to move toward a point where this disease is no longer as mysterious—or as devastating—as it is today.