Introduction
Peste des Petits Ruminants, commonly known as PPR or goat plague, represents one of the most economically devastating viral diseases affecting small ruminant farming across Africa, the Middle East, and Asia. This highly contagious disease has the potential to cause high mortality rates reaching as high as 90 percent in susceptible herds.
The global impact of Peste des Petits Ruminants extends beyond individual farms. Recent eradication initiatives have made significant progress, but PPR remains a critical concern in regions with limited veterinary infrastructure and vaccination programs.
ass=”yoast-text-mark” />>This comprehensive guide explores every aspect of Peste des Petits Ruminants disease in goats and sheep, from the underlying viral mechanisms to practical disease management, diagnostic approaches, and evidence-based preventive measures. Whether you’re a veterinary student, practicing veterinarian, or commercial livestock producer, this article provides detailed, scientifically-grounded information to help you recognize, manage, and prevent this serious disease.
What Is Peste des Petits Ruminants (PPR)?
Peste des Petits Ruminants disease is a highly infectious viral infection that selectively targets small ruminants, including domestic goats, sheep, and certain wild species. The name itself—”Peste des Petits Ruminants”—originates from French, literally meaning “plague of small ruminants,” which accurately describes the devastating nature of the infection within susceptible populations.
The disease manifests as an acute to subacute condition characterized by fever, multiple mucosal lesions, gastrointestinal dysfunction, and respiratory complications. PPR disease in goats tends to be more severe than in sheep, with young animals (kids and lambs) experiencing higher mortality rates than adults. In naive populations with no previous exposure to the virus, the disease can spread with alarming speed, affecting 80 to 100 percent of animals within a herd in just a few weeks.
What makes PPR particularly challenging is its rapid spread through direct contact, its capacity to overwhelm the immune system, and the secondary bacterial infections that frequently develop during the course of illness. Despite advances in veterinary medicine, there remains no specific antiviral cure for Peste des Petits Ruminants, making prevention through vaccination the cornerstone of any effective disease control strategy.
Etiology: Understanding the PPR Virus
The Viral Classification and Structure
Peste des Petits Ruminants virus belongs to the genus Morbillivirus within the family Paramyxoviridae. This viral family includes other significant pathogens such as measles virus in humans, distemper virus in dogs, and rinderpest virus in cattle. This virus belongs to a group called morbilliviruses, which also includes the viruses that cause measles in humans and rinderpest in cattle. The Morbillivirus classification indicates that PPRV shares fundamental biological characteristics with these related viruses, including its structure, replication mechanism, and ability to suppress host immune responses.
The PPRV particle is relatively small, measuring approximately 150 to 300 nanometers in diameter. The virus possesses a lipid bilayer envelope studded with two critical proteins: the hemagglutinin (H) protein and the fusion (F) protein. These surface proteins are responsible for viral attachment to host cell receptors and subsequent cell membrane fusion, allowing the virus to penetrate and infect target cells.
The viral genome consists of a single strand of negative-sense RNA approximately 15.9 kilobases in length. This RNA encodes six structural proteins and several nonstructural proteins that orchestrate the viral replication cycle. Unlike DNA viruses, RNA viruses like PPRV have inherent limitations in error-checking during replication, leading to naturally high mutation rates. This characteristic has implications for vaccine development and explains why the virus can generate antigenic variants over time.
Viral Strain Variation
Four distinct genotypes of Peste des Petits Ruminants virus have been identified and characterized: genotype I (Asia and Middle East), genotype II (West Africa), genotype III (East and Central Africa), and genotype IV (West Africa and central regions). While all strains cause disease in both goats and sheep, they demonstrate varying degrees of virulence and geographic distribution.
Genotype I is predominantly found in the Middle East and Asian regions, including historical occurrence in India, Turkey, and surrounding areas. Genotype II, isolated primarily from West African countries, has shown historical association with more severe disease manifestations. Genotypes III and IV are more prevalent in sub-Saharan African regions, with genotype III particularly common in East African nations. The geographic distribution of these strains has important implications for regional disease management and vaccine strain selection.
Viral Stability and Transmission Characteristics
Peste des Petits Ruminants virus demonstrates moderate environmental stability, surviving for extended periods in nasal and oral secretions. The virus can persist for several hours on contaminated surfaces, particularly on bedding, feeding equipment, and water troughs. This environmental persistence is considerably less than that of some other viral pathogens, but it remains significant enough that contaminated equipment and facilities can serve as transmission vectors.
The virus exhibits greater stability in cool, moist environments compared to warm, dry conditions. In tropical climates with high temperatures and low humidity, environmental transmission becomes less likely, whereas temperate regions with moderate temperatures provide more favorable conditions for viral survival. This environmental stability plays a crucial role in understanding seasonal patterns of Peste des Petits Ruminants outbreaks in different geographic regions.
Host Susceptibility and Species Affected
Primary Susceptible Species
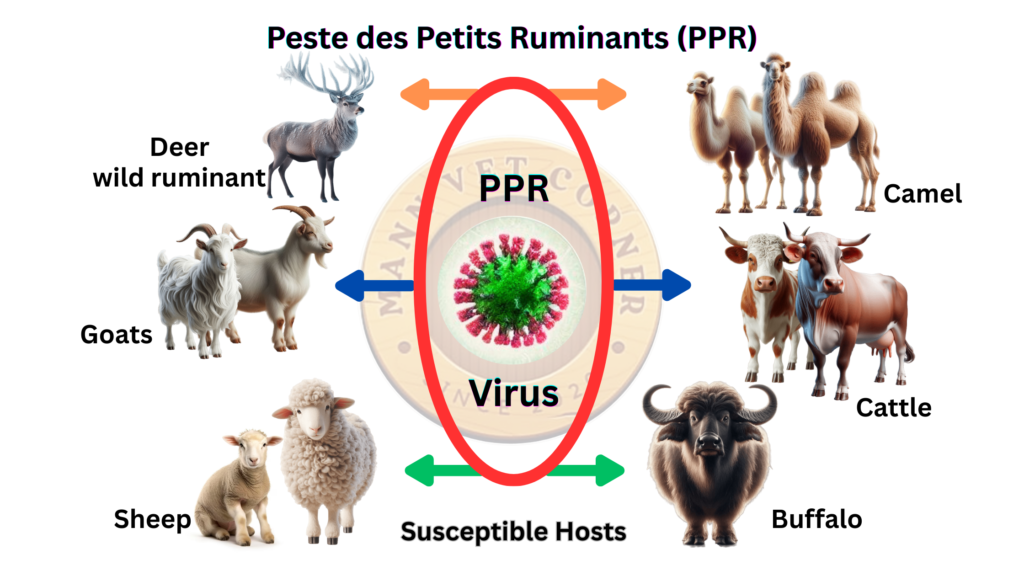
The animals most at risk from PPR are domestic sheep and goats. Domestic goats represent the most severely affected species in Peste des Petits Ruminants infections. Goats exhibit profound susceptibility with high mortality rates, clinical signs of considerable severity, and efficient viral shedding. The disease in goats progresses rapidly, with many animals deteriorating significantly within just 7 to 10 days of infection.
Domestic sheep also demonstrate significant susceptibility, though typically with somewhat milder clinical presentations compared to goats. Sheep may show similar fever and systemic signs but often develop less severe mucosal ulceration and diarrhea. Despite milder initial presentations, sheep still experience considerable mortality under severe infection conditions, particularly in young lambs.
Young animals—kids (baby goats) and lambs—consistently show higher mortality and more severe disease manifestations compared to adult animals. This age-related susceptibility relates to the incomplete development of the young animal’s adaptive immune system. Animals younger than three months of age face substantially elevated mortality risk, sometimes exceeding 80 to 90 percent when exposed to field strains of Peste des Petits Ruminants virus.
Secondary Susceptible Species
Beyond the primary targets of domestic goats and sheep, numerous wild species have demonstrated susceptibility to Peste des Petits Ruminants virus. These include various wild ungulates such as African buffalo (Cape buffalo), wildebeest, gazelles, impalas, and ibex. These wild species can contract infection from domestic herds, develop varying degrees of clinical disease, and potentially serve as virus reservoirs.
The severity of disease in wildlife generally ranges from subclinical infections to severe systemic illness, depending on the species and viral strain involved. Some wildlife species, particularly African buffalo, can develop chronic infections and serve as persistent virus sources, complicating disease eradication efforts in regions with significant wild ruminant populations. This wildlife dimension adds complexity to Peste des Petits Ruminants control programs in areas with shared grazing lands between domestic and wild species.
Non-Susceptible Species
Despite experimental demonstrations of PPRV infection in cattle, pigs, and certain other species under laboratory conditions, these animals do not develop clinical disease and do not transmit the virus under natural circumstances. Cattle, despite their relatedness to goats and sheep, prove resistant to clinical manifestation of Peste des Petits Ruminants. This species barrier is fortunate for regions with mixed cattle and small ruminant herds, as it prevents PPR from becoming a pan-ruminant pandemic.
Dogs, cats, and other carnivorous species are not susceptible to Peste des Petits Ruminants infection. This makes it unlikely that companion animals can contract or transmit PPR, though theoretically they could serve as mechanical vectors if they contact infected secretions and subsequently interact with naive sheep or goats.
Incubation Period and Early Infection Stage
Understanding the Incubation Phase
The incubation period of Peste des Petits Ruminants represents the time interval between initial viral exposure and the appearance of observable clinical signs. For most animals exposed to PPRV, clinical manifestations become apparent between the fourth and sixth day following infection. However, considerable variation exists, with incubation periods ranging from as brief as three days to as prolonged as ten days, depending on multiple biological factors.
The length of the incubation period is influenced by several determinants, including the viral load at exposure (the concentration of infectious particles the animal contacts), the virulence of the specific viral strain involved, the route of viral entry, and the individual animal’s baseline immune competence. Animals exposed to high viral loads may develop signs more rapidly, whereas those with minimal exposure and robust pre-existing immunity might show delayed clinical manifestation.
Viral Dynamics During Incubation
During the incubation period, the PPRV virus undergoes rapid replication within host tissues, but the animal exhibits no outward signs of illness. The animal appears clinically normal, maintains normal appetite and behavior, and shows normal body temperature throughout this phase. Meanwhile, the virus is actively multiplying in lymphoid tissues and beginning its systemic spread.
Despite the absence of clinical signs, infected animals begin shedding virus during the latter portion of the incubation period. This represents a critical epidemiological challenge: animals can transmit Peste des Petits Ruminants to other animals before showing any visible signs of illness themselves. This subclinical shedding during incubation significantly complicates disease control efforts, as infected animals cannot identified and isolated based on appearance alone.
Viral shedding increases substantially once clinical signs become apparent, with peak viral loads typically occurring during the first week of clinical illness. This explains why animals in the acute phase of disease represent the highest transmission risk to other animals in the herd.
Factors Affecting Incubation Duration
Young animals, particularly kids and lambs less than three months of age, frequently develop clinical signs more rapidly than older animals, sometimes showing fever and initial signs within three to four days. This accelerated progression in young animals partly reflects their incompletely developed immune systems and their generally higher overall metabolic rates.
Breed variations may influence incubation duration and disease severity, though all breeds remain susceptible to PPR. Some research suggests certain indigenous breeds, particularly those with longer evolutionary exposure to PPR in endemic regions, may develop clinical signs with slightly less rapidity than purely recently introduced breeds.
Environmental stressors during the incubation period—including overcrowding, poor nutrition, concurrent parasitic infections, or other simultaneous illnesses—can shorten the incubation period by suppressing immune function and allowing more rapid viral replication.
Clinical Signs and Symptoms of Peste des Petits Ruminants
Prodromal Phase (Initial Stage)
Peste des Petits Ruminants illness begins with a prodromal phase where the animal’s general demeanor changes noticeably. Affected animals typically present with listlessness, reduced movement, and apparent discomfort. Feed intake decreases notably, and animals may isolate themselves from the rest of the herd, showing reduced social interaction with herdmates.
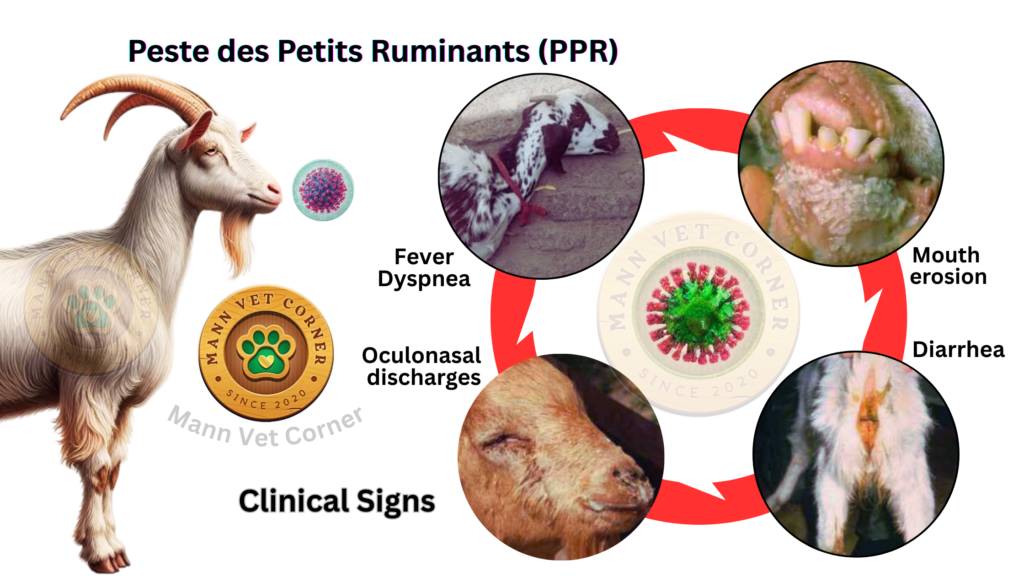
High fever develops during this initial phase, with body temperatures typically rising to 104 to 106 degrees Fahrenheit (40 to 41 degrees Celsius). In some severely affected animals, fever may exceed 107 degrees Fahrenheit. The fever frequently appears biphasic, with an initial temperature spike, brief amelioration, and then renewed fever escalation. This biphasic pattern is particularly characteristic of Peste des Petits Ruminants and helps differentiate it from certain other febrile conditions.
Affected animals show signs of systemic illness, including muscle trembling, reluctance to stand or move, and generalized weakness. The eyes appear sunken and the mucous membranes become congested, appearing redder than normal. The animal may exhibit tachycardia (elevated heart rate) and tachypnea (rapid breathing).
Ocular and Nasal Discharge Phase
Within one to two days of fever onset, characteristic ocular and nasal discharges appear. Initial eye discharge is typically watery and copious, causing obvious wetness around the orbital region. The nasal discharge similarly begins as a serous (watery) secretion but progressively becomes more mucoid, then increasingly purulent (pus-containing) as the disease progresses.
These ocular and nasal secretions are infectious, containing high concentrations of viable PPRV particles. The discharge coats the face and surrounding hair, and when animals rub their faces against feeding equipment or their own bodies, they contaminate these surfaces with infectious secretions. This mucosal discharge is a primary vehicle for disease transmission between animals and represents why nose-to-nose contact and shared feeding equipment become significant transmission routes.
The ocular congestion can become quite severe, with conjunctivitis developing. Some severely affected animals develop periorbital edema (swelling around the eyes) and may show photophobia (light sensitivity), preferring to remain in shaded areas.
Oral and Mucosal Lesion Development
One of the most characteristic features of Peste des Petits Ruminants disease is the development of erosions and ulcerations throughout the oral cavity. These lesions typically appear on the tongue (dorsal and ventral surfaces), the gingiva (gums), the hard palate, and the buccal mucosa (inner cheeks). The lesions begin as tiny red dots that rapidly progress to distinct ulcerations, often with a grayish-white necrotic center surrounded by inflamed, bleeding tissue.
The development of oral lesions in PPR occurs in a predictable sequence. Initially, petechiae (pinpoint hemorrhages) appear, followed by erosions of the superficial epithelium. Within a short timeframe, frank ulcerations develop that often coalesce, creating larger areas of mucosal damage. In severe cases, extensive sloughing of the oral mucosa can occur.
These oral lesions cause considerable pain, explaining why affected animals dramatically reduce their food intake. Animals may attempt to eat but frequently drop food from the mouth. Salivation increases markedly as animals drool excessively, with saliva sometimes blood-tinged from the damaged mucosal surfaces. The mouth may develop a characteristic odor due to secondary bacterial infections of the damaged tissues.
The pain associated with oral lesions also affects drinking behavior. Animals become reluctant to drink, further contributing to dehydration. In some cases, the pain may be so severe that animals appear to have difficulty swallowing.
Respiratory Tract Involvement
Respiratory tract involvement becomes increasingly prominent as Peste des Petits Ruminants progresses. Animals develop productive coughing, initially with a harsh, dry quality that progressively becomes more wet and productive. Nasal discharge escalates, particularly the purulent component, and animals may demonstrate dyspnea (difficult or labored breathing) as the disease advances.
Pneumonia develops as a consequence of both direct viral infection of the respiratory epithelium and secondary bacterial invasion. The cough becomes more productive, sometimes with foam or mucus visible at the nares. Respiration rate increases significantly, and in severe cases, animals show obvious respiratory distress with open-mouth breathing and extended necks as they struggle for air.
Bronchopneumonia, the predominant form of respiratory infection in PPR, develops from the viral destruction of protective respiratory epithelium combined with secondary bacterial colonization. Common bacterial agents involved in secondary respiratory infections include Pasteurella multocida and Mannheimia haemolytica, both normal inhabitants of ruminant airways that proliferate when the protective mucosal barrier is compromised.
Gastrointestinal Manifestations
Diarrhea develops in the majority of Peste des Petits Ruminants cases, typically appearing within several days of fever onset. Initially, feces may become slightly soft or pasty, progressing to frank diarrhea within 24 to 48 hours. The diarrhea frequently contains mucus and blood, giving it a characteristic appearance that veterinarians and experienced farmers often recognize as highly suggestive of PPR.
The gastrointestinal involvement stems from viral damage to the intestinal epithelium, particularly affecting the small intestine and colon. The virus causes epithelial ulceration, inflammation, and disruption of normal intestinal barrier function. Secondary bacterial infections exacerbate the mucosal damage, and the resulting inflammatory response leads to massive fluid secretion into the intestinal lumen.
Severe diarrhea leads to rapid fluid loss, contributing substantially to dehydration. Animals with PPR-associated diarrhea can lose water equivalent to 10 to 15 percent of their body weight within just a few days. This fluid loss, combined with reduced water intake due to oral lesions and general malaise, results in profound dehydration in many affected animals.
Appetite suppression combines with gastrointestinal dysfunction to create a pattern of severe malnutrition during the acute illness. Animals simultaneously lose their drive to eat while their gastrointestinal tract fails to properly absorb nutrients. This dual insult to nutritional status explains the rapid weight loss and visible emaciation that develops in animals surviving the acute phase.
Systemic Signs and Disease Progression
As Peste des Petits Ruminants progresses beyond the initial phase, systemic manifestations become increasingly severe. The fever may persist at high levels, remain biphasic, or occasionally resolve temporarily, only to recur. Lymph nodes—particularly the mandibular, pharyngeal, and mesenteric nodes—enlarge noticeably.
The animal’s general condition deteriorates progressively. Visible dehydration becomes evident, with loss of skin turgor (the skin becomes tented if pinched), dry mucous membranes, and increasingly sunken eyes. The animal becomes progressively weaker, reluctant to stand, and in severe cases, may remain recumbent (lying down) for extended periods.
In terminal stages of disease, animals may develop secondary complications including aspiration pneumonia (if they inhale liquid while debilitated), septicemia (bacterial invasion of the bloodstream through damaged mucosal barriers), and multiple organ dysfunction. The combination of severe dehydration, malnutrition, respiratory compromise, and secondary infections creates conditions incompatible with survival in many severely affected animals.
Age-Related Variations in Clinical Presentation
Young kids and lambs frequently demonstrate more severe disease than adult animals. In kids less than three months of age, clinical signs tend to be more pronounced, the disease progression more rapid, and the outcome more frequently fatal. Young animals may develop severe respiratory disease with visible dyspnea within several days of fever onset, whereas adult animals might not develop severe respiratory compromise until later in the illness.
Very young neonatal animals—particularly those less than one week of age—represent a particularly high-risk group. Although these animals rarely encounter PPRV in protected production systems, those exposed suffer extremely high mortality approaching 95 to 100 percent.
Adult animals often mount a more effective immune response than young animals, and while they develop all the characteristic clinical signs of PPR, the disease may progress more slowly, allowing for a longer survival window. Some adult animals, particularly those with concurrent good nutritional status and minimal secondary infections, may recover from uncomplicated PPR, though permanent production losses remain inevitable.
Breed Differences in Clinical Expression
While all breeds remain susceptible to Peste des Petits Ruminants virus, some variations in disease manifestation have been noted. Indigenous or locally adapted breeds in endemic areas sometimes demonstrate somewhat less severe disease than recently introduced or purely non-native breeds. This likely reflects evolutionary adaptation rather than true breed resistance, as endemic populations have developed some level of acquired immunity through repeated exposures over generations.
However, these breed differences should not be interpreted as providing meaningful protection. Even indigenous breeds suffer substantial mortality when exposed to naïve populations of animals without vaccination or previous exposure.
Morbidity and Mortality Rates in Peste des Petits Ruminants
Understanding Morbidity Patterns
Morbidity—the proportion of exposed animals that develop clinical disease—in Peste des Petits Ruminants outbreaks typically ranges from 50 to 100 percent, depending on multiple factors. In naive populations with absolutely no prior exposure to the virus and no vaccination coverage, morbidity frequently approaches or reaches 100 percent. In herds with partial immunity from previous exposure or incomplete vaccination coverage, morbidity rates may be lower, ranging from 50 to 80 percent.
The rapidity of disease spread is remarkable. In a susceptible herd, the first infected animal may originate from contact with animals from an affected herd. Within just a few weeks, the disease can spread to encompass the vast majority of the herd’s animals as the virus finds naive hosts with no protective immunity.
Factors influencing morbidity in a given situation include the degree of herd immunity (whether from prior exposure or vaccination), the virulence of the circulating viral strain, the density of animals in the affected herd, the efficiency of disease transmission pathways, and the presence of other concurrent infections or stressors that suppress immune function.
Mortality Rates and Severity Patterns
Mortality from Peste des Petits Ruminants varies considerably but typically ranges from 50 to 90 percent in affected animals. This represents a mortality rate substantially higher than most other viral diseases of small ruminants. In young kids and lambs, particularly those less than three months of age, mortality frequently approaches 80 to 90 percent. Adult animals show somewhat lower mortality, typically in the 50 to 70 percent range, though severe disease and death remain common even in mature animals.
The mortality rate is significantly influenced by the virulence of the specific viral strain involved. Some viral strains circulating in particular geographic regions are notably more virulent, producing higher mortality rates even in the same host species and age group. Genotype II strains, for example, historically have been associated with higher mortality rates compared to genotype I strains.
Case fatality rate—the proportion of diagnosed/affected animals that succumb to the disease—is typically very high. In untreated animals with severe systemic disease, case fatality rates approach 70 to 90 percent. Even with supportive care and treatment, many severely affected animals do not survive, reflecting the severity of the disease and the substantial tissue damage that occurs during infection.
Economic Impact of Mortality and Morbidity
The economic consequences of Peste des Petits Ruminants outbreaks extend beyond the immediate mortality. Surviving animals experience prolonged recovery periods, during which they produce no milk and produce little or no meat weight gain. Production losses continue for months even after animals recover, as they rebuild body condition lost during severe illness.
Reproductive consequences are substantial. Pregnant animals frequently abort when infected with PPRV, representing a compounded loss of both the dam’s current productivity and the prospective offspring. Lactating dams dramatically reduce milk production during illness, directly impacting the growth of young animals dependent on that milk for nutrition.
Secondary economic impacts include the costs of veterinary care, supportive treatments, and prevention measures. In many endemic regions, however, economic constraints limit the availability of these interventions, making prevention through vaccination the most cost-effective approach.
Pathogenesis: How Peste des Petits Ruminants Develops in the Animal Body
Initial Entry and Primary Replication Sites
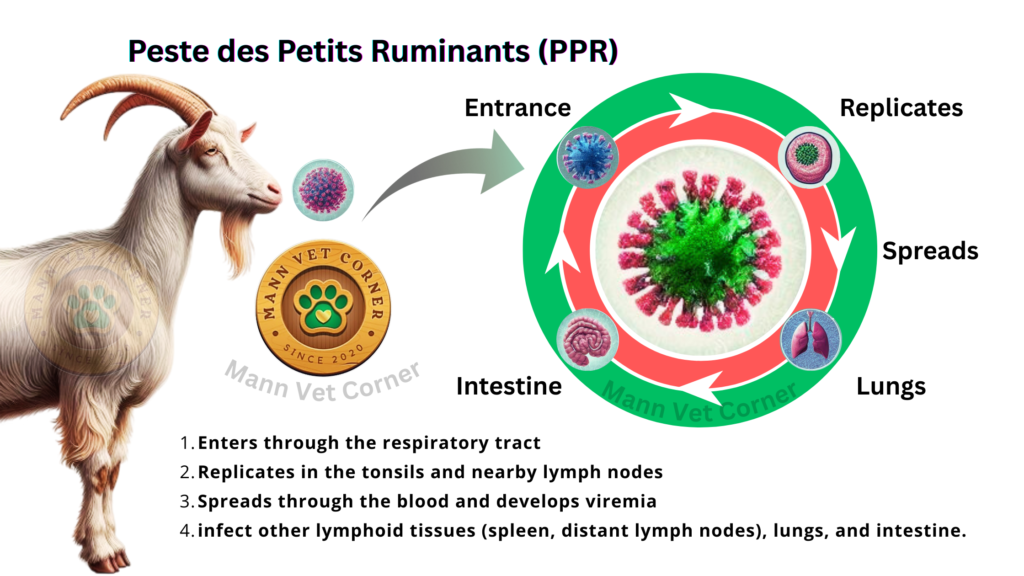
Peste des Petits Ruminants virus enters the animal body primarily through the respiratory tract, specifically via inhalation of virus-containing aerosol droplets expelled by infected animals. The virus can also enter through the oral cavity via ingestion of contaminated food, water, or environmental surfaces that have been soiled with infectious secretions. Theoretically, the virus might enter through abraded skin or mucous membranes, though these routes are considered less important under typical field conditions.
Following entry through the respiratory tract, PPRV begins its replication in the epithelial cells lining the upper respiratory passages. However, the true amplification of infection occurs when the virus reaches the lymphoid tissues associated with the respiratory tract—specifically the palatine tonsils and the pharyngeal lymph nodes. These lymphoid tissues contain abundant lymphocytes and antigen-presenting cells, creating an ideal environment for viral replication.
The tonsils and regional cervical lymph nodes become the primary replication sites during the first several days of infection. The virus infects various immune cells within these tissues, particularly B lymphocytes, T lymphocytes, and macrophages. Rather than simply infecting epithelial cells, PPRV cleverly targets the immune cells themselves, setting the stage for the profound immunosuppression that characterizes severe PPR.
Progression to Viremia and Systemic Spread
By approximately the third to fourth day of infection, viral replication becomes sufficiently prolific that infectious virions are released into the bloodstream, establishing viremia (the presence of virus in circulating blood). During viremia, the virus circulates throughout the body, delivered to virtually every tissue through the normal circulation of blood.
The viremia is frequently biphasic, meaning two separate elevations of viral titer in the blood occur, separated by a temporary decline. The first viremia wave results from primary replication in lymphoid tissues and initial escape into the bloodstream. Temporary reduction in viral titers occurs as the developing immune response begins to eliminate some circulating virus. A second viremia wave often develops as the virus successfully replicates in secondary target tissues.
During this systemic distribution phase, PPRV reaches and begins infecting cells in numerous target tissues throughout the body. The spleen, which contains abundant lymphoid cells, becomes massively infected. Lymphoid tissues throughout the body—including mesenteric lymph nodes, bronchial lymph nodes, and various other regional nodes—become sites of active viral replication.
The respiratory epithelium becomes infected as virus distributed through the bloodstream accesses lung tissue. The gastrointestinal epithelium similarly becomes infected from the circulating virus, establishing the basis for the enteritis (intestinal inflammation) that produces diarrhea. The oral mucosa becomes infected, setting up the characteristic oral ulcerations.
Immune System Dysfunction and Lymphocyte Destruction
One of the defining characteristics of Peste des Petits Ruminants pathogenesis is the profound immunosuppression that develops. PPRV preferentially infects and destroys lymphocytes, the essential cellular components of the adaptive immune system. Both B lymphocytes (which produce antibodies) and T lymphocytes (which orchestrate cellular immune responses) become infected and destroyed.
This lymphocyte destruction occurs through direct viral cytotoxicity (the virus directly kills infected cells), triggering of apoptosis (programmed cell death) in infected lymphocytes, and triggering of immune responses against infected lymphocytes that also kill uninfected bystander cells. The net result is a dramatic reduction in circulating lymphocyte numbers and in the lymphocyte populations within lymphoid tissues.
This immunosuppression explains why secondary infections with bacteria and other opportunistic pathogens become so prevalent in PPR cases. The animal’s ability to mount effective antibody responses and cell-mediated immune responses is severely compromised. Bacterial pathogens that are normally controlled by intact immune function flourish in the immunosuppressed animal.
The immunosuppression is not only profound but also appears to be relatively long-lasting. Animals recovering from severe PPR may show evidence of impaired immune function for weeks or even months after the acute infection resolves, making them susceptible to other infections during this convalescent period.
Epithelial Damage and Mucosal Ulceration
PPRV causes direct infection and destruction of epithelial cells lining the respiratory tract, gastrointestinal tract, and oral mucosa. The epithelial cells lining these surfaces are relatively specialized cells responsible for creating an impermeable barrier between internal tissues and the external environment. Viral replication within these cells causes their lysis (breaking open), creating defects in the normally protective epithelial barrier.
In the oral cavity, direct viral infection causes the distinctive ulcerations characteristic of PPR. The initial infection of the epithelial cells triggers inflammatory responses, leading to vasodilation, edema, and infiltration of inflammatory cells. The damaged, inflamed epithelium then sloughs away, creating the eroded, ulcerated surface that becomes secondarily infected with bacteria.
In the gastrointestinal tract, similar epithelial destruction occurs. The villi—the small finger-like projections that normally line the small intestine and greatly increase its surface area for nutrient absorption—become blunted and flattened as the epithelium is destroyed. This destruction dramatically reduces the intestine’s ability to absorb nutrients and also disrupts the tight junctions that normally prevent fluid from being secreted into the intestinal lumen.
The damaged intestinal epithelium becomes permeable to enteric bacteria, allowing bacterial translocation (invasion of the bloodstream by bacteria that normally remain confined to the intestinal lumen). This contributes to systemic infection and septicemia in severely affected animals.
Inflammatory Response and Tissue Damage
Beyond the direct damage caused by viral infection, the inflammatory response mounted by the animal contributes substantially to tissue injury. Affected tissues develop intense inflammatory infiltration as the immune system attempts to control the infection. This inflammatory response, while necessary to fight the infection, causes considerable collateral damage to tissue architecture.
In the lungs, the inflammatory response creates bronchopneumonia—an inflammatory consolidation of lung tissue that impairs gas exchange. In the intestines, inflammation contributes to the diarrhea through multiple mechanisms: increased intestinal secretion of fluid in response to inflammatory mediators, reduced epithelial barrier function, and increased intestinal motility driven by inflammatory stimuli.
The inflammatory response also drives fever production. Viral infection and immune cell activation release pyrogens—fever-producing substances—that act on the hypothalamic thermoregulatory center, resetting the body temperature set point to higher levels. The resulting fever, while part of an adaptive host response (fever can slow viral replication), contributes to metabolic demands and further stresses the already-compromised animal.
Dehydration and Electrolyte Disturbances
The severe diarrhea characteristic of PPR causes massive fluid losses from the body. The damaged intestinal epithelium cannot properly reabsorb the large volumes of fluid secreted into the intestinal lumen in response to inflammation and infection. Additionally, reduced fluid intake due to anorexia (reduced appetite) and dysphagia (difficulty swallowing related to oral lesions) limits fluid replacement.
This fluid loss leads to dehydration—a reduction in total body water content that impairs numerous physiological processes. As dehydration progresses, plasma volume decreases, reducing the blood volume available for circulation. Circulating blood viscosity increases, requiring greater cardiovascular effort to maintain tissue perfusion.
Concurrent with fluid loss, electrolyte disturbances develop. The watery diarrhea characteristic of viral enteritis contains substantial quantities of sodium, potassium, chloride, and bicarbonate. The loss of these essential electrolytes disrupts multiple physiological processes. Hypokalemia (low blood potassium) develops and can impair cardiac function and exacerbate muscle weakness.
Metabolic acidosis frequently develops in animals with severe PPR, resulting from both the loss of bicarbonate in diarrhea and the production of lactic acid during periods of inadequate tissue perfusion. This acidosis further compromises cardiovascular function and contributes to the animal’s deteriorating condition.
Secondary Bacterial Infections
The profound immunosuppression combined with damaged epithelial barriers creates conditions favorable for secondary bacterial infections. Bacteria that would normally be held in check by intact immune defenses flourish and multiply excessively.
In the respiratory tract, secondary bacterial infections develop as the viral destruction of respiratory epithelium removes the primary defense barrier. Pasteurella multocida and Mannheimia haemolytica, bacteria that normally colonize ruminant airways without causing disease in immune-competent animals, proliferate extensively. These bacteria cause bronchopneumonia superimposed on the viral pneumonia, dramatically worsening respiratory disease.
In the gastrointestinal tract, the damaged intestinal epithelium permits bacterial translocation, with enteric bacteria invading tissues and entering the bloodstream. Septicemia and endotoxemia (systemic effects of bacterial endotoxins) develop, contributing substantially to shock and organ failure in terminal cases.
Secondary bacterial infections of oral ulcers occur, deepening the tissue damage and potentially extending into deeper tissues (creating oral abscesses). The bacteria produce additional inflammatory mediators that worsen the systemic inflammatory response.
Nutrient Malabsorption and Metabolic Dysfunction
Beyond the direct loss of calories due to reduced food intake, animals with PPR suffer from severe malabsorption due to the damaged intestinal epithelium and reduced absorptive surface area. Proteins, lipids, carbohydrates, and micronutrients that would normally be efficiently absorbed by intact intestine are instead lost in diarrhea.
The animal’s metabolic rate increases in response to fever and infection, substantially raising caloric demands just as nutrient intake and absorption decline dramatically. The combination of increased metabolic demands, reduced intake, and impaired absorption creates a severe negative energy balance.
Protein catabolism accelerates, as the body breaks down its own muscle tissue to release amino acids for gluconeogenesis (glucose production) and immune cell production. This explains the rapid development of muscle loss and emaciation even in animals surviving the acute infection.
Organ Dysfunction and Terminal Events
In the most severe cases, systemic infection and organ dysfunction progress to multiple organ failure and death. Septicemia results in endotoxin release and excessive inflammatory mediator production, causing septic shock. Cardiovascular decompensation develops as hypovolemic shock (shock due to inadequate circulating volume) worsens progressively.
Acute kidney injury develops in some severely affected animals, resulting from hypoperfusion during severe dehydration and shock. Hepatic dysfunction may develop from hypoperfusion, direct infection, or from the effects of sepsis. Respiratory failure progresses as pneumonia worsens and gas exchange becomes increasingly inadequate.
The terminal event in most severely affected animals is either septic shock or respiratory failure, or in some cases, a combination of multiple organ failures developing concurrently.
Necropsy Findings: Post-Mortem Examination Results
Oral Cavity Lesions
Upon necropsy examination of animals that died from Peste des Petits Ruminants, the oral cavity typically displays the characteristic lesions that experienced veterinarians recognize as highly suggestive of PPR. Erosions and ulcerations are evident on the dorsal and ventral surfaces of the tongue, the gingiva (gums), the hard palate, and the buccal mucosa (inner cheeks and lips).
These ulcerations typically measure several millimeters in diameter, though extensive lesions may coalesce into larger areas of tissue loss. The ulcerated surfaces may demonstrate varying degrees of necrosis (tissue death), with some ulcers showing dark, necrotic centers and margins of inflamed, congested tissue. In chronic cases or cases complicated by secondary bacterial infection, ulcers may demonstrate a grayish-white purulent material.
In some cases, a characteristic “cheesy” or leathery exudate may coat the surfaces of ulcers, representing a combination of necrotic epithelial cells, fibrin (a clotting protein), and secondary bacterial infection products. The oral tissues surrounding obvious ulcers typically appear congested and edematous (swollen).
The extent of oral lesions varies considerably, ranging from relatively subtle erosions appreciable only upon careful examination to extensive, confluent ulcerations affecting large surfaces of the oral cavity. Severe oral lesions extending into deeper tissues may show penetration into the underlying oral mucosa or even the muscular tissues beneath.
Gastrointestinal Tract Findings
The gastrointestinal tract in animals dying from PPR demonstrates characteristic changes reflecting the severe viral and bacterial gastroenteritis that occurred during the animal’s illness. The stomach frequently shows congestion (abnormal accumulation of blood) of the mucosa, and in some cases, small erosions or ulcerations similar to but typically less extensive than those in the mouth.
The small intestine typically appears congested, with the mucosa showing edema (swelling). In many cases, the intestinal contents demonstrate the characteristic bloody, mucoid diarrhea that was observed during the animal’s life. The intestinal mucosa may show focal areas of hemorrhage, and in cases with extensive mucosal ulceration, frank areas of mucosal loss are evident.
A particularly characteristic finding in PPR is the appearance of the colon and cecum. These organs frequently demonstrate a distinctive “zebra striping” pattern—alternating bands of congestion and normal coloration. This striping results from the congestive changes present in some portions of the colonic mucosa while other regions remain less severely affected. This zebra striping pattern is sufficiently characteristic that it’s often used as a confirmatory finding in diagnostic necropsy.
In some cases, the cecum and colon contain fibrin (clotted material) and hemorrhagic exudate, indicating the severe inflammatory response that occurred in these regions. The mesenteric lymph nodes (nodes associated with the intestines) frequently show enlargement and congestion, consistent with the severe enteritis.
Lymphoid Tissue Changes
The lymph nodes are almost universally enlarged in animals that died from PPR. The cervical lymph nodes (neck nodes), mesenteric nodes (intestinal nodes), and bronchial nodes (lung-associated nodes) all typically demonstrate marked enlargement. These nodes may show varying degrees of congestion, with the cut surface often appearing dark and friable (easily crumbling).
Edema surrounding enlarged lymph nodes is frequent, indicating the inflammatory swelling that occurred. In some cases, hemorrhage within lymph nodes is evident, suggesting intense inflammatory activity.
The spleen in PPR cases typically shows marked enlargement (splenomegaly) and congestion. The cut surface appears dark red and the organ may be quite friable, indicating severe inflammation and tissue damage. Splenic involvement reflects the substantial viral replication that occurred within this lymphoid organ.
Peyer’s patches (areas of lymphoid tissue in the intestinal wall) frequently show enlargement and may demonstrate areas of hemorrhage, confirming the intensity of the enteritis that occurred.
Respiratory Tract Findings
The lungs in severe PPR cases demonstrate consolidation—areas where normal lung tissue has been replaced by inflammatory exudate. The consolidated regions typically appear firm rather than normal spongy texture, and the cut surface does not yield the normal frothy fluid. Instead, consolidated areas may exude a purulent (pus-containing) or fibrinopurulent fluid.
The consolidated lung tissue typically appears discolored—pale, gray, or darker red compared to normal lung tissue. These changes are consistent with bronchopneumonia, the predominant form of respiratory infection in PPR deaths.
In severe cases with significant respiratory involvement, edema fluid may accumulate in the airways, and the trachea (windpipe) may contain frothy fluid with a reddish tinge, indicating pulmonary edema with some hemorrhage.
Pleural effusion (fluid accumulation in the space surrounding the lungs) may be present in some cases, adding to the respiratory compromise. The pleural fluid may appear turbid or hemorrhagic depending on the degree of inflammation.
Additional Organ Involvement
The liver may appear congested and enlarged (hepatomegaly), reflecting the hepatic involvement that frequently occurs in septicemia and systemic infection. The liver may show multifocal areas of necrosis (tissue death) if bacterial septicemia occurred.
The kidneys may demonstrate congestion and possibly show evidence of acute tubular necrosis if renal failure developed during the terminal stage of illness. Fluid in the renal pelvis may be present, indicating dehydration.
In cases where rupture of the intestinal wall occurred (as can happen with extensive ulceration and perforation), peritonitis (abdominal inflammation) may be evident, with fibrin coating the abdominal organs and peritoneal surface.
Absence of Pathognomonic Changes
It is important to note that while the combination of findings above is highly suggestive of PPR, no single pathognomonic (uniquely characteristic) finding is pathognomonic for PPR when taken in isolation. Other viral diseases can cause oral ulceration, diarrhea, and respiratory disease. Additionally, animals dying from secondary complications may not show all the typical PPR changes.
Definitive diagnosis requires laboratory confirmation, typically through viral isolation, PCR detection of viral nucleic acid, or immunohistochemical identification of PPRV antigen in tissues.
Diagnostic Approaches for Peste des Petits Ruminants
Clinical Diagnosis and Limitations
Experienced veterinarians in endemic areas can frequently presume a diagnosis of PPR based on characteristic clinical signs and necropsy findings. The distinctive combination of high fever, oral ulceration, diarrhea with zebra striping pattern, respiratory disease, and lymphoid enlargement is highly suggestive of PPR when it occurs in a young small ruminant in an endemic area.
However, clinical diagnosis alone is insufficient for definitive confirmation, as other diseases can produce similar clinical presentations. Differential diagnoses include contagious ecthyma (orf), foot-and-mouth disease, rinderpest (in cattle), and various other infections.
Virus Isolation
Virus isolation remains a classic diagnostic approach, though it is less commonly used in modern diagnostic laboratories. Nasal secretions, oral swabs, or blood collected during the acute phase of illness can be cultured in susceptible cell cultures. PPRV grows relatively well in several cell culture systems, including kidney cells from various species and fetal fibroblast cells.
Growth of PPRV in cell culture produces characteristic cytopathic effects (visible cell damage), including cell rounding, detachment from the culture surface, and formation of syncytia (multinucleated giant cells). However, virus isolation is time-consuming, taking several days to weeks for results, and requires skilled laboratory personnel and appropriate facilities.
RT-PCR and Nucleic Acid Detection
Reverse transcription polymerase chain reaction (RT-PCR) has become the gold standard diagnostic method for Peste des Petits Ruminants. This technique detects and amplifies the viral RNA genome, providing highly sensitive and specific diagnosis. Various RT-PCR assays have been developed, including conventional RT-PCR, real-time/quantitative RT-PCR (qRT-PCR), and multiplex assays that can simultaneously detect PPR and distinguish it from related viruses.
Real-time RT-PCR provides both qualitative confirmation of viral presence and quantitative determination of viral load (the amount of virus present in the sample). This provides valuable information about the intensity of infection.
Samples suitable for RT-PCR include nasal swabs, oral swabs, blood, feces, and tissues (in necropsy cases). For optimal sensitivity, samples are best collected during the acute phase of illness when viral titers are highest, though virus can still be detected during the late acute and early convalescent phases.
Serological Diagnosis
Serological testing detects antibodies against PPRV that develop as an animal’s immune system responds to infection. These tests include ELISA (enzyme-linked immunosorbent assay), hemagglutination inhibition assays, and other antibody-detection methods.
Antibodies typically appear 7 to 10 days after infection, making serological tests useful for diagnosing animals recovering from infection or for epidemiological surveys of herd immunity. However, serological tests are not useful during the acute phase of illness when antibodies have not yet developed.
Serological tests are also useful for distinguishing vaccinated animals from naturally infected animals in vaccination programs, as certain vaccine strains produce different serological responses that can be differentiated through competitive ELISA assays.
Immunohistochemical Staining
Immunohistochemical techniques can identify PPRV antigen in tissues collected at necropsy. Tissue samples, particularly from lymphoid tissues, lung, or intestine, are processed into microscopic slides. Antibodies against PPRV are applied, and a detection system highlights areas where viral antigen is present. This allows visualization of infected tissues under the microscope.
This technique is particularly useful for necropsy diagnosis and can provide information about which tissues were most heavily infected.
Electron Microscopy
Transmission electron microscopy can visualize the virus particles directly. Samples containing high concentrations of PPRV can be examined, and the characteristic size and morphology of morbillivirus particles can be identified. However, this requires expensive equipment and highly skilled personnel, so it’s rarely used for routine diagnostics.
Treatment Options and Supportive Care
Absence of Specific Antiviral Therapy
Unlike certain viral diseases, no specifically effective antiviral drugs exist for treating Peste des Petits Ruminants virus infection. Drugs such as ribavirin, which show some activity against certain RNA viruses, have not proven clinically effective in PPR treatment. This reflects the fundamental challenge of treating RNA viral infections—the rapid viral mutation, integration into cell membranes, and ability to suppress host immune responses make them inherently difficult to treat pharmacologically.
The absence of specific antivirals means that treatment must focus entirely on supportive care—maintaining the animal’s physiological functions while the immune system attempts to clear the infection naturally.
Fluid and Electrolyte Replacement
Fluid therapy represents the cornerstone of supportive treatment for PPR. The severe diarrhea and reduced water intake create profound dehydration that requires aggressive fluid replacement. Multiple approaches to fluid therapy exist, with the choice depending on the severity of dehydration and the clinical setting.
Oral fluid therapy using rehydration solutions (similar to those used for diarrhea in human patients and calves) can be effective in animals with moderate dehydration and continuing appetite. These solutions contain appropriate proportions of sodium, potassium, glucose, and other electrolytes to replace those lost in diarrhea while promoting intestinal reabsorption of fluid.
Intravenous fluid therapy is necessary for severely dehydrated animals, animals unable to drink, or animals with shock. Polyionic crystalloid solutions (solutions with electrolyte composition matching plasma) such as lactated Ringer’s solution or normal saline are administered intravenously. The rate and volume of fluid administration must be carefully calculated based on the degree of dehydration, ongoing losses, and the animal’s cardiovascular status.
Plasma transfusions may be beneficial for animals with severe protein loss and ongoing enteritis, as they provide both fluid volume and albumin and other plasma proteins necessary for maintaining colloid osmotic pressure.
Nutritional Support and Appetite Stimulation
Animals with severe oral lesions frequently have difficulty or are unwilling to consume normal feed due to pain. Providing soft, finely ground feed or feeding pelleted rations is necessary. In some cases, hand-feeding small amounts of soft feed may encourage eating.
Nutritional support should continue throughout recovery, as animals recovering from severe PPR require substantial caloric intake to rebuild lost muscle and condition. Young animals are particularly at risk of permanent growth stunting if nutritional support during and after the acute illness is inadequate.
Vitamin supplementation, particularly B vitamins and fat-soluble vitamins, may be beneficial in severely affected animals, as the damaged intestinal epithelium will have impaired nutrient absorption even as feeding resumes.
Antibiotic Therapy for Secondary Infections
While antibiotics cannot treat the viral infection itself, they are valuable for controlling secondary bacterial infections. Broad-spectrum antibiotics with activity against respiratory pathogens (such as Pasteurella multocida and Mannheimia haemolytica) should be initiated, as secondary pneumonia is a major cause of mortality in PPR cases.
Appropriate antibiotic choices include:
- Oxytetracyclines: These broad-spectrum agents have been the traditional choice in developing countries where PPR is endemic, partly due to their availability and moderate cost. Oxytetracycline can be administered intravenously, intramuscularly, or orally.
- Penicillin-aminoglycoside combinations: Penicillin combined with gentamicin provides good coverage against common respiratory pathogens. This combination is highly effective but expensive, limiting its use in resource-limited settings.
- Fluoroquinolones: Agents such as enrofloxacin provide excellent tissue penetration and broad-spectrum activity, though cost may limit their use.
- Third-generation cephalosporins: Cephalosporins provide good activity against common secondary pathogens, though these are expensive.
Antibiotic selection should ideally be guided by bacterial culture and sensitivity testing when possible, though this may not be practical in field situations. In endemic areas, local knowledge of common secondary pathogens and their antibiotic sensitivities provides useful guidance.
Antipyretic and Anti-inflammatory Therapy
Fever management can improve animal comfort and reduce metabolic demands. Non-steroidal anti-inflammatory drugs (NSAIDs) such as phenylbutazone, flunixin meglumine, or aspirin provide both antipyretic (fever-reducing) and analgesic (pain-relieving) effects. These agents reduce the inflammatory response and associated pain from oral lesions and gastrointestinal inflammation.
Corticosteroids are sometimes used in severe cases to suppress excessive inflammatory responses, though these agents carry the risk of further immunosuppression. In moderately affected animals, the benefits of reducing inflammation may outweigh the immunosuppressive effects, but in severely immunocompromised animals, corticosteroids are potentially counterproductive.
Pain Management
Animals with severe oral ulceration and abdominal pain require adequate analgesia. In addition to NSAIDs, local topical treatments of oral ulcers with antiseptic or soothing solutions may provide symptomatic relief. Some practitioners apply topical agents containing benzocaine or other local anesthetics to oral ulcers to reduce pain during eating.
Environmental Management
Affected animals should be housed separately from healthy animals in clean, well-ventilated accommodations. Overcrowding should be avoided, as stress exacerbates illness and increases viral shedding. Soft bedding is important, as severely ill animals remaining recumbent are at risk of pressure ulcers.
Moderate ambient temperature (neither excessively hot nor cold) reduces metabolic stress. Affected animals should be protected from wind and drafts, which can worsen respiratory disease.
Duration of Treatment and Prognosis
The supportive care described above should be continued throughout the acute phase of illness and into early convalescence. However, even with aggressive treatment, mortality remains high in severely affected animals, particularly kids and young lambs. The mortality rate even with treatment typically ranges from 50 to 80 percent in affected animals, compared to 80 to 90 percent in untreated animals.
Most animals that are going to die from PPR do so within 10 to 14 days of infection. Animals surviving this period have a reasonable probability of eventual recovery, though recovery may require several weeks and complete recovery to pre-infection productivity may not occur.
Prevention: The Paramount Strategy
The economic and welfare costs of treating PPR outbreaks make prevention through vaccination the by-far most cost-effective strategy. Even in the most developed countries with access to sophisticated veterinary services, preventing PPR through vaccination is far more cost-effective than treating affected animals.
Prevention of Peste des Petits Ruminants: Vaccination Strategies
History and Development of PPR Vaccines
The first successful PPR vaccine, the Nigeria 75/1 strain, was developed decades ago and remains widely used today. This vaccine is a live-attenuated (weakened) vaccine, meaning that the virus has been genetically modified through repeated passage through cell culture to reduce its virulence while maintaining immunogenicity (ability to stimulate immune response).
Modern vaccine development has produced additional vaccine strains, including tissue culture-adapted strains and recombinant vaccines produced using genetic engineering techniques. These newer vaccines may offer certain advantages, including reduced neurovirulence and improved safety profiles, though the classical Nigeria 75/1 strain remains the most widely used worldwide due to its proven efficacy and relatively low cost.
Mechanism of Vaccine Protection
Live-attenuated PPR vaccines work by stimulating a complete immune response to PPRV without causing clinical disease. The weakened virus infects the vaccinated animal’s lymphoid tissues (particularly regional lymph nodes and tonsils) and triggers replication, but the reduced virulence of the vaccine strain prevents the severe immunosuppression and tissue damage characteristic of virulent PPRV infection.
This infection stimulates both humoral immunity (antibody production) and cellular immunity (cytotoxic T cell responses). The antibodies produced against the vaccine virus recognize virulent PPRV, providing protection against infection. The cellular immunity provides long-lasting protection and helps clear infection if virulent virus subsequently exposes the vaccinated animal.
Vaccine Administration and Regimens
Most PPR vaccines are administered by subcutaneous or intramuscular injection. The dose recommended varies somewhat among vaccines but typically ranges from 1,000 PCCID₅₀ (fifty percent tissue culture infectious dose) per animal for most vaccines.
Age-appropriate vaccination strategies maximize vaccine efficacy. In endemic areas, animals should be vaccinated starting at 3 to 4 months of age, when maternally-derived antibodies have waned sufficiently to allow vaccine responsiveness. In disease-free areas being protected, vaccination can be administered at slightly older ages.
A single dose of live-attenuated vaccine typically provides protection for at least 4 to 5 years, and often provides much longer-lasting immunity. Some countries recommend annual or bi-annual booster vaccination in very high-risk situations, though most vaccination recommendations involve vaccination every 3 to 5 years.
Young kids (less than 3 to 4 months of age) frequently possess maternally-derived antibodies from immune dams. These passive antibodies can interfere with vaccination response. For this reason, vaccination programs typically delay vaccination until these maternal antibodies have waned, typically at 3 to 4 months of age.
Efficacy and Immune Response
Properly administered live-attenuated PPR vaccines produce protection against virulent PPRV infection in 95 to 100 percent of vaccinated animals. Animals vaccinated as kids typically have high-level protection by 2 to 3 weeks post-vaccination that persists for several years.
The immune response to vaccination can be monitored through serological testing. Animals vaccinated with live-attenuated vaccine develop serum-neutralizing antibodies that are indistinguishable from antibodies produced by natural infection. This creates a challenge for distinguishing recently vaccinated animals from naturally infected animals, though newer serological assays (particularly competitive ELISA) can differentiate vaccine-induced from infection-induced antibodies in some cases.
Vaccine Adverse Events
Live-attenuated PPR vaccines are generally very safe, with serious adverse events being uncommon. Mild local reactions at the injection site (temporary swelling or heat) occur occasionally. Systemic reactions such as mild fever or transient lethargy are rare.
The vaccine virus is excreted in nasal and oral secretions of recently vaccinated animals. While the vaccine virus is substantially less virulent than wild-type virus, recently vaccinated animals theoretically could transmit vaccine virus to unvaccinated animals. In practice, vaccine-virus transmission is rare and causes either subclinical infection or mild disease at most.
Some countries have established official protocols restricting movement of recently vaccinated animals to prevent potential vaccine-virus spread, though the actual risk appears minimal in most situations.
Strategic Vaccination Programs
Different regions and countries pursue different vaccination strategies based on their PPR status. In endemic regions where PPR is regularly present, regular vaccination of all young animals provides a rolling immunity that prevents disease outbreaks. In areas attempting to eliminate PPR, vaccination is combined with rapid response to any suspected cases, movement restrictions, and sometimes depopulation of infected herds.
The FAO and OIE (World Organization for Animal Health) have developed global strategies for progressive control and eventual eradication of PPR. These strategies emphasize structured vaccination programs targeting all animals in endemic areas, movement controls to prevent viral spread, surveillance to detect outbreaks, and rapid response protocols.
Combination Vaccination Programs
In regions where multiple small ruminant diseases are present, combination vaccines targeting PPR along with other diseases (such as contagious ecthyma, foot-and-mouth disease, and others) are increasingly available. These combination vaccines reduce the number of injections required and improve overall herd health through broader disease protection.
Epidemiology and Geographic Distribution
Endemic Regions and Outbreak Dynamics
Peste des Petits Ruminants is present in Africa (affecting most sub-Saharan countries), the Middle East, South Asia, and parts of Southeast Asia and Central Asia. The geographic range of the disease has actually expanded during recent decades, likely reflecting increased livestock movement and trade.
Within endemic regions, the disease shows varying patterns of occurrence. Some areas experience regular seasonal outbreaks, while others have more sporadic occurrence. Seasonal patterns often reflect changes in animal density and management practices during certain times of year (such as rainy seasons when animals congregate around limited water sources).
Transmission and Spread Mechanisms
Direct transmission between animals through nasal and oral secretions represents the primary transmission route. Infected animals shed virus most abundantly during the acute phase of illness, with highest concentrations in nasal secretions, ocular discharges, saliva, and diarrheal feces.
Indirect transmission through contaminated equipment, facilities, or food and water sources occurs, though is generally less efficient than direct transmission. The moderate environmental stability of PPRV means contaminated environments can remain infectious for days to weeks, particularly in cool, moist conditions.
Animal movement represents a major factor in disease spread. The livestock trade (movement of animals from endemic areas to disease-free areas) has repeatedly introduced PPR into new geographic regions. Uncontrolled movement of animals between herds and regions readily spreads the disease.
Factors Promoting Outbreaks
Overcrowding, poor sanitation, nutritional deficiencies, and concurrent infections all predispose to more severe PPR outbreaks. Stress factors such as prolonged transport, weaning, or climatic extremes increase susceptibility.
Animals with compromised immune systems (from previous infections, malnutrition, or parasitic infections) develop more severe disease and shed more virus, perpetuating outbreaks.
Surveillance and Monitoring
Active surveillance systems in some regions monitor for PPRV through regular serum surveys, fecal monitoring, or clinical surveillance. Passive surveillance relies on disease reporting by farmers and veterinarians. International organizations coordinate global surveillance to track PPR occurrence and guide control efforts.
Differential Diagnosis: Distinguishing PPR from Similar Diseases
Contagious Ecthyma (Orf)
Contagious ecthyma shares some features with PPR but is readily distinguished. This disease is primarily affects the lips, muzzle, and sometimes the hooves, with characteristic proliferative lesions rather than ulcerations. The lesions of contagious ecthyma have a scabby, crusty quality compared to the wet ulcerations of PPR oral lesions. Contagious ecthyma does not typically cause the systemic signs, fever, and diarrhea characteristic of PPR.
Foot-and-Mouth Disease (FMD)
Foot-and-mouth disease causes vesicular (blister-like) lesions rather than the deeper ulcerations of PPR. FMD lesions typically affect the feet (interdigital spaces and sole) more prominently than the mouth, whereas PPR primarily affects the oral cavity. The general systemic signs of fever and malaise are present in both, but FMD causes much less gastrointestinal involvement and diarrhea than PPR.
Rinderpest (in Cattle and Related Species)
Rinderpest disease, now eradicated, was similar to PPR but affected cattle rather than primarily small ruminants. The clinical signs were similar to PPR, with fever, discharges, oral ulceration, and diarrhea. Fortunately, rinderpest no longer exists, eliminating this differential concern in current practice.
Goat Pox and Sheep Pox
Pox viruses cause systemic disease with fever and discharges but produce characteristic pustular (pus-containing) skin lesions rather than the mucosal ulcerations and zebra striping diarrhea characteristic of PPR. Pox diseases lack the profound diarrhea and gastrointestinal involvement that characterize PPR.
Caprine Arthritis-Encephalitis (CAE) and Maedi-Visna
These retroviral infections cause chronic disease with progressive signs of arthritis and neurological dysfunction. They lack the acute fever, mucosal ulceration, and diarrhea of PPR. The timeline of disease development is much more prolonged (months to years) compared to the acute 10-12 day course of PPR.
Mycoplasmal and Bacterial Pneumonia
These infections cause respiratory disease and fever but lack the mucosal ulcerations, zebra striping diarrhea, and the profound systemic involvement characteristic of PPR. These bacterial/mycoplasmal diseases develop more insidiously without the acute fever of PPR.
Parasitic Diarrhea and Gastroenteritis
Parasitic infections cause diarrhea and may affect growth and productivity, but do not produce the high fever, systemic illness, and mucosal ulcerations of PPR. Microscopic fecal examination reveals parasitic infection.
Johne’s Disease (Paratuberculosis)
This disease causes chronic, progressive diarrhea but lacks the acute fever, mucosal ulceration, respiratory disease, and rapid progression of PPR. Johne’s develops insidiously over months to years, contrasting with the acute disease course of PPR.</p>

thank you for sharing with us, I conceive this website really stands out : D.
I used to be very pleased to find this internet-site.I wished to thanks to your time for this glorious learn!! I definitely enjoying each little bit of it and I’ve you bookmarked to check out new stuff you weblog post.