Introduction
Every time you pour a glass of milk, fry an egg, or slice into a steak, you trust that food on your plate is safe. But here is a truth that most people never hear: traces of veterinary antibiotics, antiparasitic drugs, and antimicrobials may be hiding inside those very foods. These invisible contaminants — called drug residues in animal-derived food products — are not just a laboratory curiosity. They connect directly to one of the deadliest threats the world faces today: antimicrobial resistance (AMR).
This guide breaks down everything you need to know — from what drug residues actually are, why they end up in your food, how they damage your health, and what veterinarians, farmers, and consumers can do right now to stop the problem at its source.
What Are Drug Residues in Animal-Derived Food?
Drug residues refer to traces of veterinary medicinal products that remain inside the edible tissues, milk, or eggs of food-producing animals after treatment. These residues include:
- Parent compounds: the original drug molecule itself.
- Metabolites: breakdown products the animal’s body creates after processing the drug.
- Associated impurities: contaminants from drug formulations.
Common drug classes that leave residues include antibiotics (penicillin, tetracyclines, sulfonamides), antiparasitics (benzimidazoles, ivermectin), hormones, and pesticides. Each of these carries a distinct risk profile when humans consume them — even at sub-therapeutic, trace-level doses.
| Quick Fact: Global antibiotic use in livestock accounts for over 70% of total antibiotic consumption worldwide, according to WHO estimates. Much of this use is unnecessary or preventive rather than therapeutic — a direct pipeline for residue contamination. |
6 Root Causes of Drug Residues in Meat, Milk, and Eggs
Understanding why drug residues enter the food chain is the first step toward stopping them. These six causes drive the majority of contamination cases reported globally.
1. Poor Record-Keeping of Treated Animals
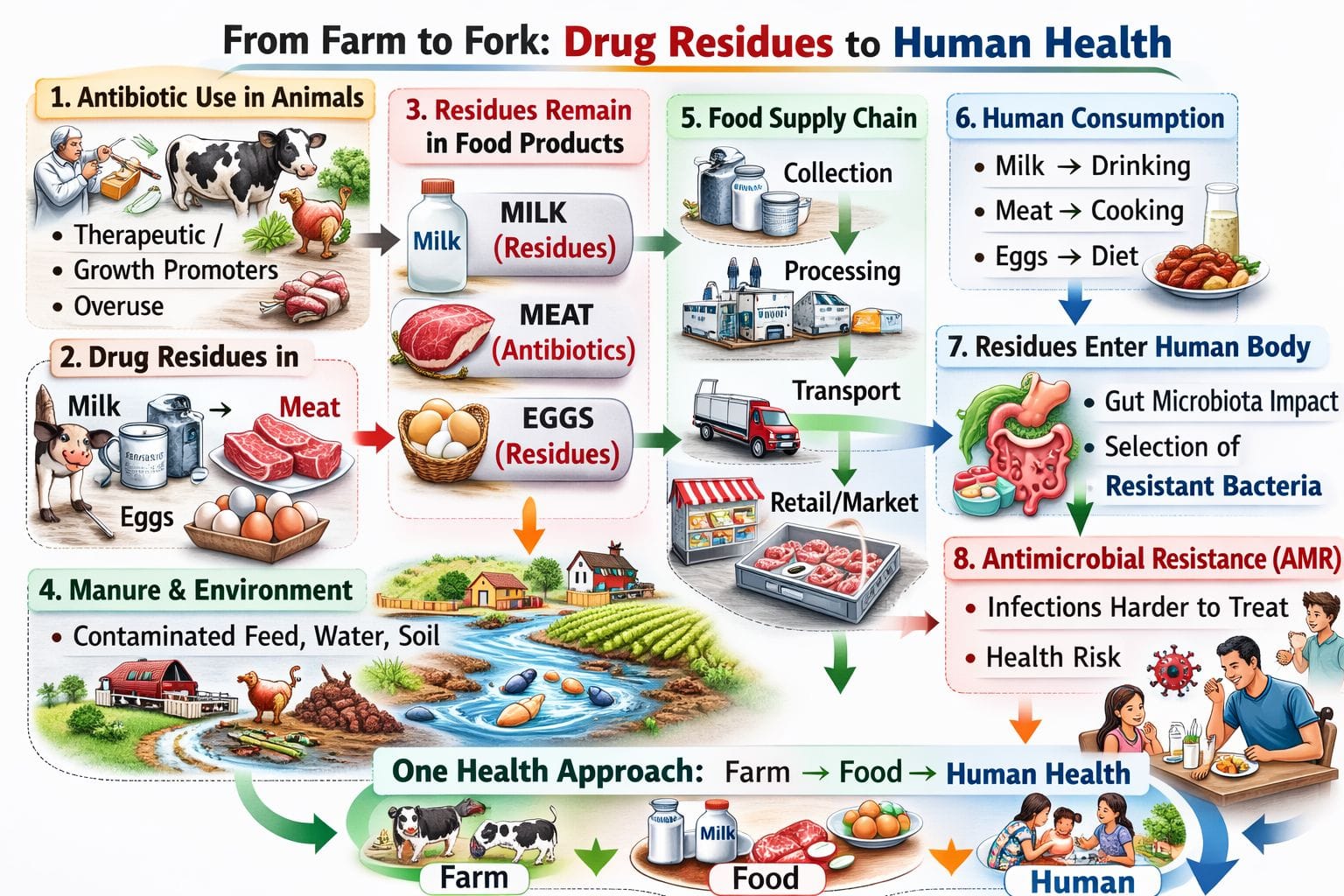
When farmers fail to track which animals received treatment, which drug was used, the exact dose administered, and the date of the last dose — they set themselves up for accidental contamination. Without written records, producers cannot accurately calculate when the withdrawal period ends. Animals enter slaughter or continue producing milk and eggs while drug concentrations in their tissues are still above safe limits.
Digital farm management systems now make record-keeping easier and cheaper than ever. There is no valid excuse for skipping it — the public health cost is simply too high.
2. Withdrawal Period for Antibiotics in Livestock
Every veterinary drug carries a withdrawal period — the minimum time between the last dose and when the animal’s products become safe for human consumption. This is not an arbitrary bureaucratic number. It is calculated based on pharmacokinetic data: how long the drug and its metabolites take to drop below the Maximum Residue Limit (MRL) established by regulatory agencies.
When producers rush animals to slaughter early, sell milk before the withdrawal period ends, or simply ignore the timeline, drug concentrations in food stay dangerously high. This single factor drives a large proportion of residue violations detected globally.
3. Extra-Label Drug Use Without Veterinary Oversight
Extra-label drug use means giving an animal a drug in a way that differs from the manufacturer’s approved label — using it in a different species, at a different dose, through a different route, or for a different disease indication. In many cases, extra-label use is legitimate and necessary. However, without expert veterinary guidance, it dramatically increases residue risk because the standard withdrawal period may not apply.
A higher dose than the label recommends means longer clearance time. A different route of administration (for example, oral instead of intramuscular injection) changes drug absorption and distribution patterns. Without recalculating withdrawal periods for the specific situation, contamination becomes almost inevitable.
4. Over-the-Counter Access to Antimicrobials Without Prescription
In many low- and middle-income countries, antibiotics meant for livestock are freely available at local markets and feed stores without any veterinary prescription. Untrained farm workers and smallholder farmers self-medicate animals based on guesswork, neighbor advice, or social media recommendations — often using the wrong drug, wrong dose, or wrong duration.
This pattern not only produces drug residues in food but also creates ideal conditions for the development and spread of antibiotic-resistant bacteria. Restricting antimicrobial access to licensed veterinary prescriptions is one of the most impactful policy changes governments can make.
5. Lack of Farmer and Consumer Awareness
Many smallholder producers genuinely do not understand that treating a cow with antibiotics and selling her milk the next morning is dangerous. They see the milk, which looks and smells normal, and conclude it is safe. Consumer awareness works in tandem — when buyers accept and purchase animal products without demanding safety certification or residue testing, market pressure to comply with regulations disappears.
Consumer education campaigns and clear food labeling policies are not soft policy extras — they are essential levers that shape producer behavior at the farm level.
6. Inadequate Veterinary Infrastructure in Rural Areas
When qualified veterinarians are not accessible — due to geographic distance, cost, or shortage of trained professionals — farmers make medication decisions alone. They rely on outdated knowledge, community norms, or packaging instructions that they may struggle to read or interpret. Building veterinary extension services into rural agricultural systems is foundational to residue prevention.
How Antibiotic Residues Cause Antimicrobial Resistance
Antimicrobial resistance develops when bacteria evolve mechanisms to survive drug exposure. Sub-therapeutic antibiotic concentrations — precisely the kind found in food residues — are among the most potent evolutionary drivers of resistance. Here is the mechanism:
When animals consume antibiotics and those drugs are not fully cleared before products enter the food chain, the residues expose the bacteria living in the human gut to low, chronic doses of antibiotics. Low doses do not kill all bacteria — they kill the susceptible ones and leave behind the resistant strains. Those resistant strains then multiply, exchange resistance genes with other bacteria via horizontal gene transfer, and establish themselves in the human microbiome.
The resulting superbugs — bacteria resistant to multiple antibiotics — cause infections that standard treatment protocols cannot cure. The WHO classifies AMR as one of the top ten global public health threats. Conservative estimates suggest resistant infections already cause at least 1.27 million deaths annually worldwide, a number that experts warn will climb sharply without aggressive intervention.
| AMR in Numbers: By 2050, AMR could cause 10 million deaths per year — surpassing cancer as the leading cause of mortality globally, according to the landmark O’Neill Commission Report on Antimicrobial Resistance. |
The chain from farm to fork to resistance is real, measurable, and preventable. Drug residues in animal foods are not just a food safety issue — they are a critical node in the global AMR network.
6 Serious Health Effects of Drug Residues on Human Consumers
1. Antimicrobial Resistance (AMR) — The Silent Pandemic
As described above, regular low-level antibiotic exposure through contaminated food selects for resistant bacteria in the human gut. This means that when a person later develops a serious infection and needs antibiotics to survive, those drugs may no longer work. The consequences range from prolonged illness to death.
2. Allergic and Hypersensitivity Reactions
Beta-lactam antibiotics — particularly penicillin and cephalosporins — cause severe allergic reactions in a significant portion of the population. Even trace residues in meat or dairy can trigger reactions in sensitized individuals. Symptoms range from skin rashes, itching, and hives to life-threatening anaphylaxis requiring emergency epinephrine. People with known penicillin allergies face genuine risk from contaminated animal products, even without knowing they consumed residues.
3. Carcinogenic Effects from Specific Drug Classes
Certain veterinary drugs carry carcinogenic properties when their residues accumulate in human tissue over time. Nitrofurans and nitroimidazoles — once widely used antimicrobials in poultry and aquaculture — produce metabolites that damage DNA and increase risk for cancers including colorectal and liver cancer. For this reason, most high-income countries have banned these drugs in food-producing animals entirely. However, enforcement remains inconsistent in many regions.
4. Disruption of Gut Microbiome Balance
The human gut hosts trillions of beneficial bacteria that regulate digestion, train the immune system, and protect against pathogens. Antibiotics such as streptomycin, tylosin, metronidazole, and vancomycin — when present as residues in food — can disturb this delicate microbial ecosystem. The result is gut dysbiosis: a state where harmful bacteria gain dominance over beneficial ones. Symptoms include chronic diarrhea, bloating, increased susceptibility to infections like C. difficile, and potentially long-term immune dysregulation.
5. Teratogenic Risks for Women
A women during pregnancy and their developing fetuses face particular danger from certain drug residue classes. Benzimidazoles, a widely used class of dewormers in livestock, are highly embryotoxic during early pregnancy. Exposure through food residues during the critical first trimester may cause congenital malformations including skeletal abnormalities, neural tube defects, and developmental delays. A women are rarely warned about this pathway of exposure.
6. Economic and Trade Consequences
Beyond direct health effects, drug residues carry devastating economic consequences. When trading partners detect residue violations, they trigger immediate export bans and product recalls — destroying entire market relationships overnight. The European Union, United States, and major Gulf markets maintain strict Maximum Residue Limit (MRL) testing at borders. A single contamination incident can cost an exporting country millions of dollars in lost trade and years of damaged reputation.
How to Detect Drug Residues in Animal Food Products
Food safety agencies and laboratories use a layered detection strategy — rapid screening to flag potential violations, followed by confirmatory tests for precise identification and quantification.
Rapid Screening Methods
Screening tests process large volumes of samples quickly and cheaply. Their goal is to sort clean samples from suspicious ones that warrant further analysis. Common approaches include microbial inhibition tests — where antibiotic residues in milk prevent bacterial growth in a test medium — and lateral flow immunoassay strips, similar in design to home pregnancy tests, that detect specific drug families within minutes. These methods give yes/no answers rather than exact concentrations.
ELISA: High-Sensitivity Immunoassay Testing
Enzyme-Linked Immunosorbent Assay (ELISA) relies on precise antigen-antibody binding reactions to detect and semi-quantify specific drugs. ELISA kits exist for dozens of antibiotic families and can detect residues at parts-per-billion concentrations. Laboratories use them for systematic monitoring programs across large sample populations. However, ELISA results occasionally produce false positives, which is why positive screens must be confirmed with more specific methods.
Gold Standard: LC-MS/MS Confirmatory Analysis
Liquid Chromatography coupled with Triple Quadrupole Mass Spectrometry (LC-MS/MS) is the gold standard for confirmatory drug residue analysis. It simultaneously identifies the exact chemical structure of a residue and quantifies its concentration with parts-per-trillion precision. No other available technique matches this combination of specificity and sensitivity. This is the method that courts, regulators, and international trade bodies require for enforcement actions.
Biosensors and Emerging Technologies
Biosensor-based detection platforms are rapidly advancing. These devices use biological recognition elements — antibodies, aptamers, or enzymes — coupled with electrochemical or optical transducers to generate readable signals when target drugs are present. Portable biosensors suitable for farm-gate and processing-plant use are entering the market, enabling real-time residue monitoring before products leave the production facility. This field will likely transform food safety testing over the next decade.
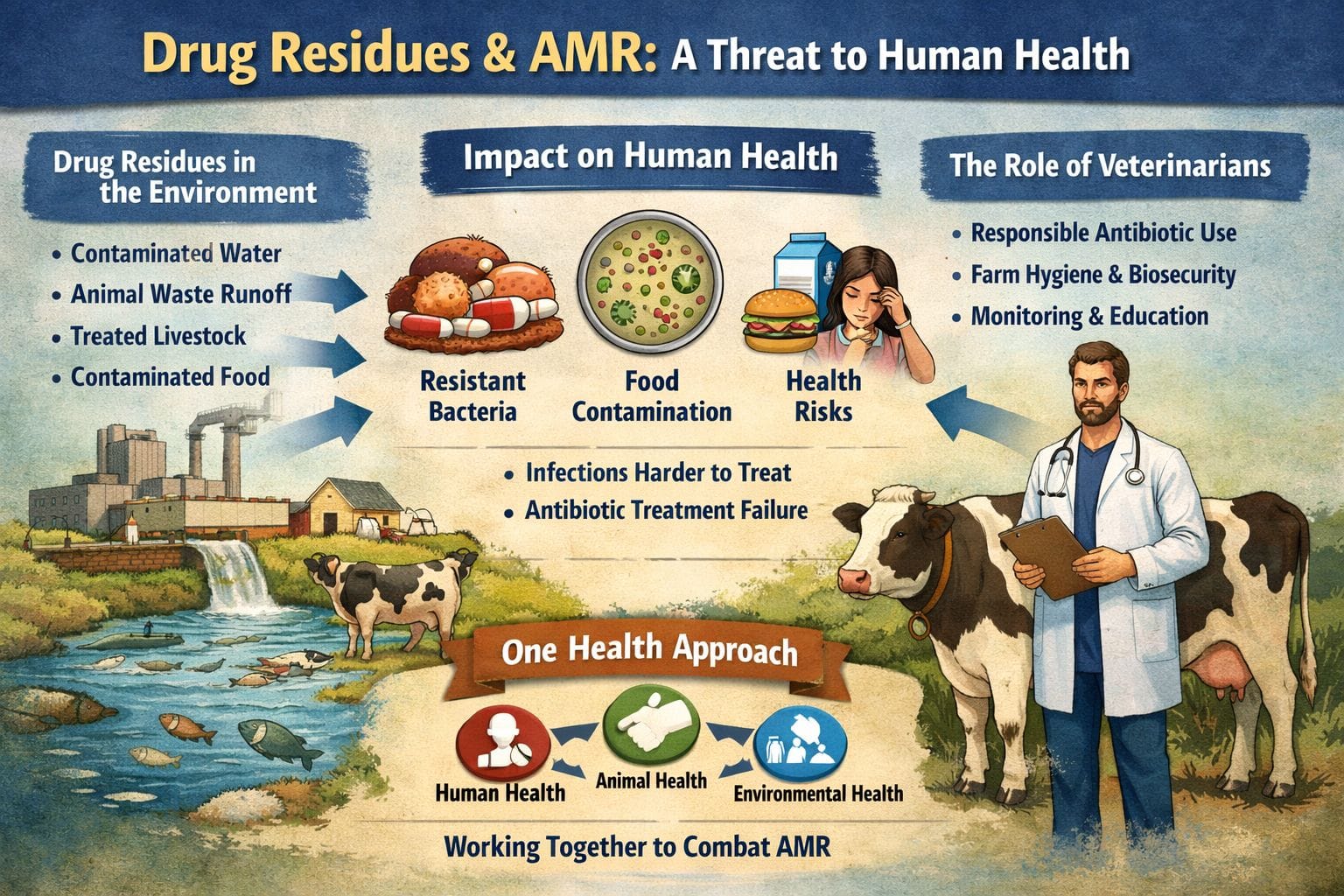
Veterinarians’ Critical Role in Preventing Antibiotic Residues in Animal Products
Veterinarians sit at the center of the drug residue problem — and its solution. As the professionals who prescribe, administer, and oversee drug use in food-producing animals, they carry unique authority and responsibility in keeping the food supply safe.
Prescribe Responsibly — Antibiotic Stewardship in Practice
Antibiotic stewardship means prescribing antibiotics only when clinical evidence justifies their use, choosing the narrowest-spectrum drug effective for the target pathogen, using the correct dose and duration, and following — or extending — withdrawal periods when extra-label use is necessary. Veterinarians who adhere to stewardship principles directly reduce both residue contamination and antimicrobial resistance selection pressure.
Communicate Withdrawal Periods Clearly to Farmers
Knowing the correct withdrawal period is not enough — veterinarians must communicate it to producers in a language they understand, confirm they have recorded it properly, and follow up to ensure compliance. This communication is a professional obligation, not optional guidance.
Educate Producers on Record-Keeping and Animal Identification
Every treated animal must be individually identified and documented. Veterinarians should train farmers on ear tagging, record-keeping apps, or paper logbooks — whatever system fits their operation — and verify that records are being maintained correctly during farm visits.
Promote Disease Prevention Over Reactive Treatment
The best antibiotic is the one never used. Veterinarians who prioritize vaccination programs, biosecurity protocols, optimized nutrition, and housing conditions that reduce disease transmission significantly cut antibiotic usage across their client farms. Healthier animals need fewer drugs — and produce fewer residues.
Support Use of Disease-Resistant Breeds
Selective breeding programs producing animals with stronger innate disease resistance offer long-term structural solutions. Veterinarians who participate in or recommend these programs contribute to farms that are fundamentally less dependent on pharmaceutical intervention.
Explore Evidence-Based Traditional Alternatives
Where scientific evidence supports their safety and efficacy, veterinarians can incorporate botanical extracts, essential oils, probiotics, and other non-antibiotic alternatives into treatment and prevention protocols. This approach reduces reliance on synthetic antimicrobials and the residue risks they carry.
Maximum Residue Limits (MRLs): The Regulatory Line Between Safe and Unsafe
Regulatory agencies worldwide establish Maximum Residue Limits (MRLs) — the highest legally permitted concentration of a drug residue in a food product. The Codex Alimentarius Commission, established by the WHO and FAO, sets internationally recognized MRLs that serve as the basis for trade agreements.
MRLs are not calculated arbitrarily. They are derived from toxicological data, including Acceptable Daily Intake (ADI) values established through animal studies, factored against average food consumption patterns for the target population. A food product testing above the MRL for any drug residue is legally non-compliant and subject to removal from sale, recall, and potential criminal penalties against producers.
However, MRL compliance alone does not guarantee zero health risk. Regulatory frameworks are built around average exposures for average consumers. Individuals who eat animal products above average quantities, women with infants, people with drug allergies, and immunocompromised patients may face meaningful risk even from residues within MRL limits — a reality that drives ongoing scientific debate about whether current standards are protective enough.
The Global AMR Action Plan: Where Drug Residue Control Fits In
The WHO Global Action Plan on Antimicrobial Resistance, endorsed by all UN member states, identifies five strategic objectives. Reducing agricultural antibiotic use and drug residues in food is central to Objective 3: optimizing the use of antimicrobial medicines in human and animal health.
Countries implementing One Health frameworks — which address AMR simultaneously across human medicine, veterinary medicine, agriculture, and environmental sectors — show the strongest results in bending the AMR curve downward. Drug residue control programs are a core pillar of every effective national AMR strategy.
The global food system is too integrated for any country to solve this alone. Meat, dairy, and eggs cross international borders daily. Resistant bacteria hitchhike along the same routes. Residue standards, testing requirements, and veterinary prescription rules need harmonization across trading partners — or the weakest link will continue to undermine progress everywhere else.
What Consumers Can Do to Reduce Their Exposure to Drug Residues
Individual consumer choices create market-level pressure that drives producer behavior. Here are practical steps every consumer can take:
- Buy certified products: Look for antibiotic-free, hormone-free, or organic certification labels — these require third-party auditing and residue testing beyond standard regulatory requirements.
- Diversify your protein sources: Rotating between plant proteins, wild-caught seafood, and certified animal products reduces cumulative exposure to any single contaminant class.
- Ask questions at the point of sale: Farmers’ markets, butchers, and direct-from-farm channels often provide full transparency on drug use and withdrawal period compliance.
- Advocate for policy change: Support legislation requiring prescription-only antibiotic access for livestock, mandatory residue testing at processing plants, and publicly accessible testing results.
Conclusion: Drug Residues and AMR Are Solvable — But Only Together
Drug residues in animal-derived foods are not an inevitability of modern agriculture. They are the predictable result of specific, identifiable failures: poor record-keeping, ignored withdrawal periods, uncontrolled drug access, inadequate veterinary oversight, and low consumer awareness. Every one of these failures is fixable.
The connection between drug residues and the global AMR crisis is not theoretical — it is biochemical, epidemiological, and urgent. Resistant bacteria do not respect international borders, income levels, or political systems. They travel in food, in water, and in people. Stopping their spread requires every actor in the food system — veterinarians, farmers, processors, regulators, and consumers — to take their specific piece of the problem seriously.
Veterinarians who practice rigorous antibiotic stewardship, farmers who follow withdrawal periods without exception, governments that enforce MRL standards and restrict over-the-counter antimicrobial sales, and consumers who demand transparent, certified food — together, they form a chain of accountability strong enough to produce real change.
The world does not have to accept a future where routine infections become untreatable. But building a different future requires acting now, at every level of the food system. The plate in front of you is a good place to start.
Frequently Asked Questions (FAQs) About Drug Residues in Animal Food
Q: Are drug residues in food products dangerous in small amounts?
Yes. Even trace amounts can trigger allergic reactions in sensitive individuals, and chronic low-level exposure to antibiotic residues drives the selection of resistant bacteria in the gut microbiome — a process with serious long-term health consequences.
Q: How long does it take for antibiotics to leave an animal’s system?
This varies widely by drug, species, dose, and route of administration. Withdrawal periods range from 24 hours for some drugs to several weeks for others. Some drugs — like certain sulfonamides in egg-laying hens — have withdrawal periods measured in multiple weeks. Always refer to the specific drug label and consult a licensed veterinarian.
Q: Can cooking destroy drug residues in meat?
Cooking reduces residue concentrations in some cases, but it does not eliminate them reliably. Many drug residues are heat-stable. Some actually convert to more toxic forms when heated. Cooking is not a substitute for proper withdrawal period compliance.
Q: What is the difference between antibiotic-free and organic labeling?
Antibiotic-free labels indicate that no antibiotics were used during the animal’s life. Organic certification additionally requires antibiotic-free status plus adherence to organic feed, animal welfare, and environmental standards verified by accredited certifying bodies. Organic standards are generally more comprehensive but also more costly for producers.
Q: Which foods carry the highest drug residue risk?
Milk, honey, eggs, liver, and kidney tissue tend to accumulate drug residues at higher concentrations than muscle meat, because these products and organs are actively involved in drug metabolism and excretion. Testing programs globally consistently flag these matrices as priority monitoring targets.